Mechanosensing
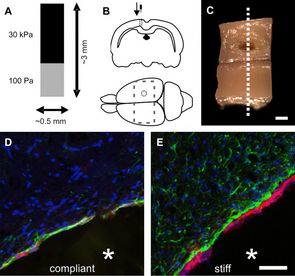
Cells actively sense and respond to a variety of mechanical signals – a process known as mechanosensing. Mechanical cues provided by the extracellular environment can modulate a wide spectrum of cellular processes, including cell proliferation, differentiation and migration.
In order to examine the impact of microenvironment stiffness on cell behaviour we use different 2D and 3D hydrogel-based systems. The main advantage of the employed gels is the possibility to independently control the mechanical and biochemical properties of the substrate. Moreover, the stiffness values obtained through tissue mechanics measurements enable us to better tune the hydrogels and perform mechanosensing studies both in physiological and non-physiological mechanical conditions.
As summarised in Franze et al. [2], we have begun to understand that mechanics plays an important role in the development, functioning and disorders of the nervous system. We have shown that CNS glial cells respond to mechanical cues in vitro and in vivo. Substrates with a stiffness far above the physiological microenvironment of the brain cause both microglia and astrocytes to display an acute and late chronic inflammatory response typical for reactive gliosis [1, 3]. Moreover, we have revealed that substrate stiffness influences oligodendrocyte progenitor cells’ survival, proliferation, differentiation and migration capacities in vitro [4]. Recently, colleagues have shown that local tissue stiffness influences neuronal growth and pathfinding even in vivo [5].
Besides neurons and glial cells, our lab also investigates the mechanosensing abilities of other cell types, including neutrophils, macrophages, adipocytes and cancer cells. For example, we have demonstrated the suitability of engineered 3D peptide-functionalised hydrogels to investigate in vitro how the interactions between cancer cells and the extracellular matrix proteins around them modulate tumour development and progression [6].
Insights gained from these projects may ultimately lead to a better understanding of physiological processes and may open novel avenues to treat the underlying degenerative pathologies such as neurodegenerative diseases or, as emphasised in Lacour et al. [7], to improve implant biocompatibility and design.
[1] P. Moshayedi, G. Ng, J. C. F. Kwok, G. S. H. Yeo, C. E. Bryant, J. W. Fawcett, K. Franze, and J. Guck, “The relationship between glial cell mechanosensitivity and foreign body reactions in the central nervous system,” Biomaterials, vol. 35, iss. 13, p. 3919–3925, 2014.
[2] K. Franze, P. a. Janmey, and J. Guck, “Mechanics in neuronal development and repair.,” Annual Review of Biomedical Engineering, vol. 15, iss. 1, p. 227–51, 2013.
[3] P. Moshayedi, L. F. da Costa, A. Christ, S. P. Lacour, J. Fawcett, J. Guck, and K. Franze, “Mechanosensitivity of astrocytes on optimized polyacrylamide gels analyzed by quantitative morphometry,” Journal of Physics: Condensed Matter, vol. 22, p. 194114, 2010.
[4] A. Jagielska, A. L. Norman, G. Whyte, K. V. J. Vliet, J. Guck, and R. J. M. Franklin, “Mechanical Environment Modulates Biological Properties of Oligodendrocyte Progenitor Cells,” Stem Cells and Development, vol. 21, iss. 16, 2012.
[5] D. E. Koser, A. J. Thompson, S. K. Foster, A. Dwivedy, E. K. Pillai, G. K. Sheridan, H. Svoboda, M. Viana, L. F. da Costa, J. Guck, C. E. Holt, and K. Franze, “Mechanosensing is critical for axon growth in the developing brain,” Nature Neuroscience, vol. 19, iss. 12, p. 1592–1598, 2016.
[6] A. V. Taubenberger, L. J. Bray, B. Haller, A. Shaposhnykov, M. Binner, U. Freudenberg, J. Guck, and C. Werner, “3D extracellular matrix interactions modulate tumour cell growth, invasion and angiogenesis in engineered tumour microenvironments,” Acta Biomaterialia, vol. 36, p. 73–85, 2016.
[7] S. P. Lacour, G. Courtine, and J. Guck, “Materials and technologies for soft implantable neuroprostheses,” Nature Reviews Materials, vol. 1, p. 16063, 2016.





