Willkommen auf der Homepage der Abteilung für biologische Optomechanik
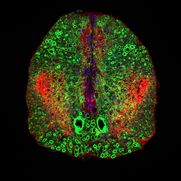
Zellen sind die grundlegenden Einheiten biologischer Systeme. Sie haben besondere physikalische Eigenschaften, die es ihnen ermöglichen, sich in ihrer physikalischen 3D-Umgebung zu bewegen und ihre biologischen Funktionen zu erfüllen. Wir untersuchen diese physikalischen - mechanischen und optischen - Eigenschaften von lebenden Zellen und Geweben mit Hilfe neuartiger photonischer und biophysikalischer Werkzeuge, um ihre biologische Bedeutung zu testen. Unser Ziel ist der Transfer unserer Erkenntnisse in die medizinische Anwendung auf den Gebieten der verbesserten Diagnose von Krankheiten und neuer Ansätze in der regenerativen Medizin.
Kontakt
Bitte setzen Sie sich bei Anfragen aller Art mit uns in Verbindung:
Abteilung Guck
Max-Planck-Institut für die Physik des Lichts
Staudtstr. 2
91058 Erlangen
guck-office@mpl.mpg.de
Tel: +49-9131-7133-501/-502
Fax: +49-9131-7133-990
Das Max-Planck-Institut hat seinen Sitz direkt am Südgelände der Friedrich-Alexander-Universität Erlangen-Nürnberg, auf dem die Technische Fakultät angesiedelt ist. Informationen zur Anfahrt finden Sie hier.