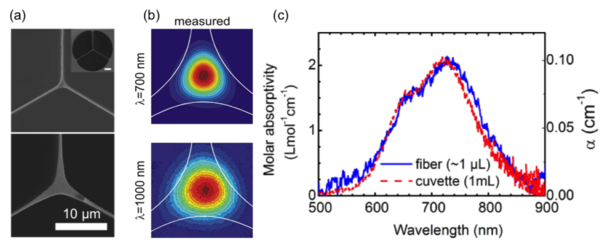
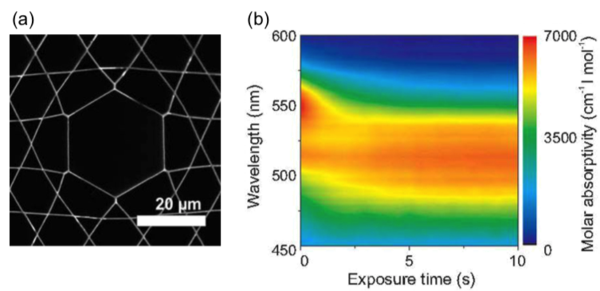
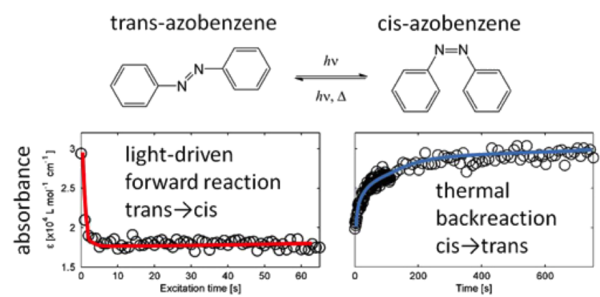
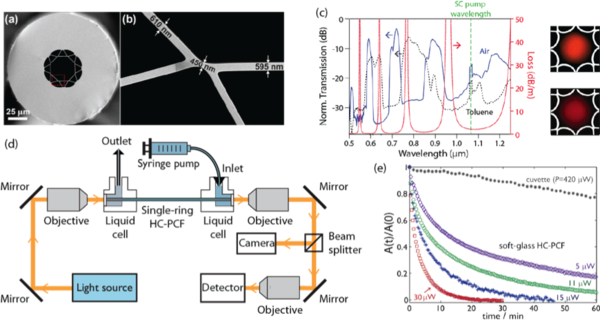
The development of new photoactivated anticancer complexes is of major importance for the effective treatment of cancer. Such complexes are inert, nontoxic and are activated only locally in cancer cells by light. In collaboration with co-workers from the University of Warwick in the UK, we have developed a new method that allows rapid testing and optimization of photochemical compounds [Chen (2010); Unterkofler (2012)]. It makes use of the unique waveguiding properties of liquid-filled hollow-core photonic crystal fibres to bring together for the first time the fields of microfluidics, chemistry, and optics. The hollow core is loaded with chemicals and acts as a miniature photochemical reactor. Both the chemicals and the excitation light are confined to the core. Consequently, the entire sample volume interacts strongly with the light, resulting in much shorter reaction times than in conventional cuvettes. Importantly, the required sample volume is reduced by four orders of magnitude. The reaction is monitored in real-time by in-fibre broadband absorption spectroscopy, offering insight in the dynamics of the chemistry. As a proof-of-principle study, the photochemical conversion of Vitamin B12a to vitamin B12b upon excitation with blue laser light was investigated. In a continuous-flow configuration, the method enables a rapid optimization of the excitation wavelength and light dosage of photoactivated anticancer complexes.